Reactions
| Prev |
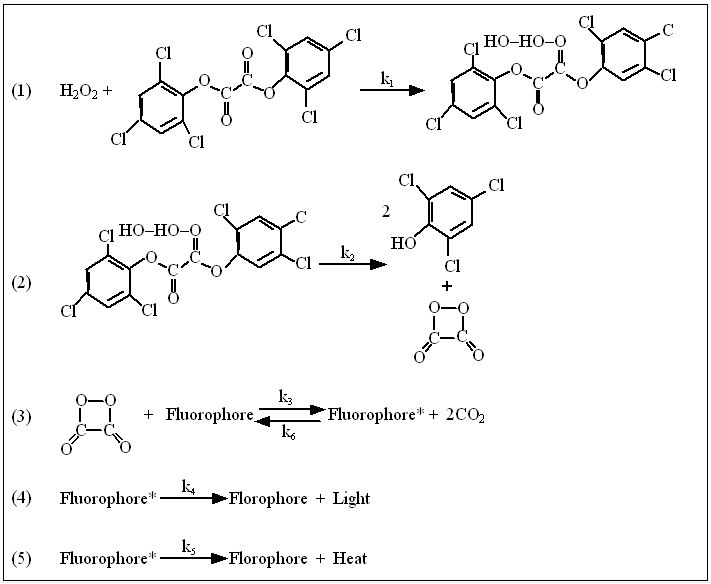
There are three compounds that are necesary for glowsticks to produce light. The first two, bis(2,4,6-trichlorophenyl)oxalate (TCPO) and hydrogen peroxide, combine to produce a complex which reacts with the third chemical, a dye (generally an anthracene derivative) and the dye emits the light.
The hydrogen peroxide oxidizes the TCPO, to form trichlorophenol and an unstable peroxyacid ester. The unstable peroxyacid ester decomposes, resulting in phenol and 1,2-dioxetanedione. The dioxetanedione complexes with the dye molecule and releases light. In Mechanism (1) the dye does not decompose or become inactive after emmitting light while in Mechanism (2) it is inactive after emitting light.
The dye releases light of a color dictated by its molecular structure. A list of common dyes and their colors follows:
Blue - 9,10-diphenylanthracene
Green - 9,10-bis(phenylethynyl)anthracene
Yellow - 1-chloro-9,10-bis(phenylethynyl)anthracene
Yellow - Rubrene
Orange - 5,12-bis(phenylethynyl)-naphthacene
Orange - Rhodamine 6G
Red - Rhodamine B
We are going to offer two mechanisms to model the glow sticks.
Mechanism (1) Full Set of Balance Equations
The reaction steps given by Chasteen have been modified to include the reverse reaction for the formation of activated flourophore.

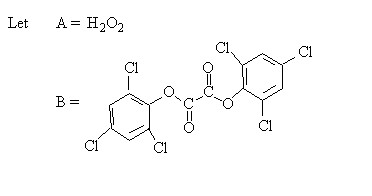
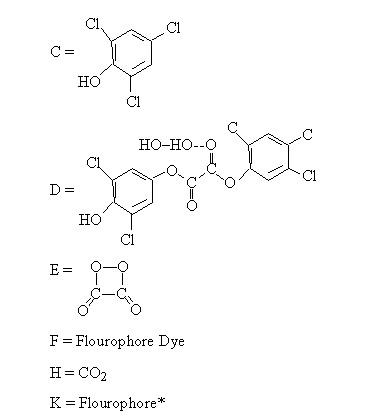
Then Mechanism (1) in notation form is

T. G. Chasteen, "The Chemiluminescence of Luminol and bis(2,4,6-trichlorphenyl)oxalate (TCPO)" [see http://www.shsu.edu/%7Echm_tgc/JPPdir/JPP1999/]
| Prev |