Release Notes Version 3.2.1
Released October 14, 2013
This update provides fixes to issues that were introduced with Release 3.2.
Release Notes Version 3.2
Released October 7, 2013
The following changes apply to all user groups:
Various enhancements to the study application and SCR application have been made in order to simplify the process of recording Subject Enrollment Numbers:
- Study Application Enhancements:
- New help text added to question 3-1.1.
- Section 8 has been updated (and new help text added) to clarify what data the IRB expects the study team to enter and to eliminate duplicate data entry.
- Question 8.1.1 has been removed. Subject enrollment totals will now automatically be calculated in question 8.2.
- SCR Application Enhancments:
- Various improvements have been made to the data table in question 2-2.1 in order to make it more user-friendly.
- A new Overages column will populate in the question 2-2.1 table if the study team enrolled more subjects than they were approved for. If this happens, or if there is a negative number present in the change since the last reporting period, question 2-2.1.1 will be required.
All references in eRRM to the Division of Research Development and Administration (DRDA) have been replced with the Office of Research and Sponsored Projects (ORSP) as per the department's name change effective January 2012.
Wording in section 7-1 and section 27 of the application has been changed to include the term "Concealment."
MCRU review will no longer be a stopping point in the review process for studies requiring MCRU resources and will occur concurrently with the ancillary committee and IRB review process. For studies requiring MCRU review, workspace messages will display to indicate if MCRU review is in progress or if MCRU review has been completed.
All study team members who have been granted edit rights on a submission will now be able to execute the applicable “Withdraw” activity when the submission is in-progress and is in an editable state by the study team. When the submission is withdrawn, an email notification will be sent to all study team members.
An error causing some Amendments requiring Ancillary Committee review to route incorrectly has been fixed.
The email group for ORSP has been updated so that they will continue to receive notifications about projects that are moved into Full or Partial Project Hold.
The following changes apply to Study Teams:
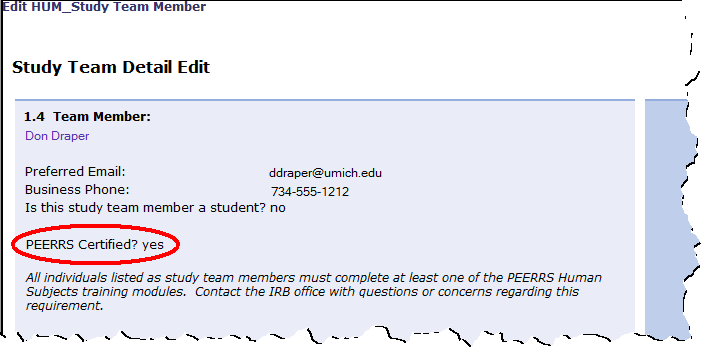
Study Team Member Details (section 1.4 of the study application) will now display a "Yes" or "No" for PEERRS Certified? Completed PEERRS certification modules will no longer display.
Section 7-2 (Special Considerations) of the study application is now available to Dearborn studies.
Once an application has moved to a state of “PRC Review Complete,” the corresponding study workspace messages have changed. The messages now read, “PRC review complete, study team action required,” and, “PRC approval issued; complete the remainder of the application and submit.”
A bug preventing the Accept Role activity from displaying when it should has been fixed.
Various changes have been made to section 42 and section 42-1 MCRU Information of the study application.
Direct links to the IND and IDE worksheets are now available in sections 15 and 16 (Drugs and Devices) of the study application.
Studies indicating UM Flint or Dearborn as the IRB can now select the Requesting Review by a Non-UM IRB application type.
Users can now search by uniqname when adding study team members.
For projects submitted to IRBMED, study teams will be asked to answer a new question on the study application: 10-1.1.1 Does the Informed Consent use the sentences required for Applicable Clinical Trials: "A description of this clinical trial will be available on www.ClinicalTrials.gov, as required by US law. This website will not include information that can identify you. At most, the website will include a summary of the results. You can search this website at any time."?
- If the study team answers "Yes" to question 10-1.1.1 or indicates the study is a clinical trial in question 1-2.7, a new Update NCT Number activity will become available, which is used to enter the required NCT number.
For study teams who indicate that research subjects will be exposed to ionizing radiation from external radiation sources (question 21.1 of the study application), a new question 21.1.1 will display asking the user to identify the source of the radiation. If Radioactive Material/Radioactive Source (sealed source, etc) is selected, they will be routed to a new section of the study application, 21-4 RDRC/SHUR Information.
Study team members not affiliated with UM will now need to answer four (4) Conflict of Interest screening questions when they accept their role on the study team. If any of the screening questions are answered “yes,” the person will need to download a paper disclosure form the COI office’s website (link provided below screening questions) and upload it in eRRM. See the (Friends Account) Accepting Your Role on the Study Team step-by-step for instructions.
The following changes apply to IRB Staff and Committee Members:
A new approval letter template for IRB-MED has been created, which is to be used in cases identified by CRAO where Medicate Part A and Medicare Part B are involved in Device studies.
The IRB Reviewer Checklist has been updated. If a management plan has been provided to the IRB, a reviewer checklist item will be generated for IRB Reviewers to indicate acceptance of the management strategy.
Question 10-1 on the Staff Reviewer Checklist has been changed to read, “Each ICD is in the proper format.”
Important dates related to AE/ORIOs have been added to the AE Summary Report and the ORIO Summary Report.
On studies for which section 25. HIPAA U-M Covered Components is required, a new item will appear on IRB Primary, IRB Secondary, and Privacy Board reviewer checklists.
An error causing duplicate auto-generated contingencies to be created on studies sponsored by the Department of Defense has been fixed.
Enhancements have been made to the Member Details tab in the Committee Workspace in order to address performance issues.
Staff Reviewer checklist items and auto-generated contingencies referencing section 8-2 of the study application have been changed to reference section 8-1. This section was renumbered in the RM 3.1 release but was not updated for reviewer checklists and contingencies.
The following changes apply to Ancillary Committees:
Formatting issues when generating the PRC approval letter have been fixed.
The following change applies to Regulatory Oversight:
The Regulatory Oversight role now has access to the Manage Documents activity on Applications. This activity is only available when there is not an amendment in progress.