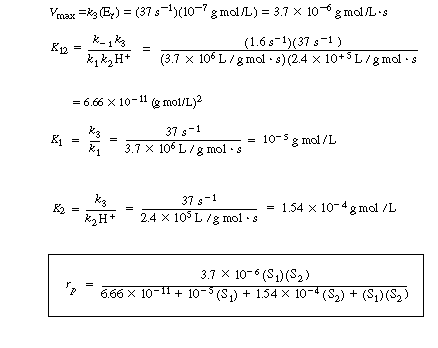Before considering the possibility of going directly from the apoenzyme to the holoenzyme, assume that the
rate of dissociation of the complex (ADH![]() NAD+),
NAD+),
![]()
is irreversible, and show that the initial rate law for ethanol in the enzyme cofactor reaction sequence discussed earlier is of the form
![]()
(RE7.4-2.1)
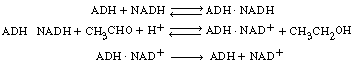
Let E = ADH, S1 = CH3CHO, S2 = NADH, ![]() = NAD+, and P1 = CH3CH2OH. Then
= NAD+, and P1 = CH3CH2OH. Then
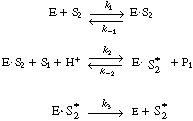
By adding the rate law for the rate of formation of ethanol (P 1),
![]()
to the equation for![]() (below), we see that the rate law for ethanol can be written as
(below), we see that the rate law for ethanol can be written as
![]()
(RE7.4-2.2)
Application of the PSSH to the holoenzyme (E![]() S2) and the apoenzyme
S2) and the apoenzyme
(E![]()
![]() )
)
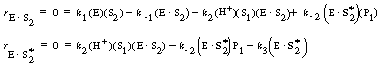
allows one to solve for the concentrations of the cofactor-enzyme complexes in terms of S2, S2*, and E.
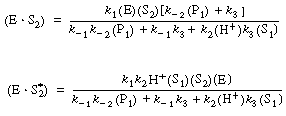
(RE7.4-2.3)
(RE7.4-2.4)