is carried out inside spherical catalyst pellets of two different sizes. The effectiveness factor for pellet size A is 0.8 and for size B it is 0.4. Catalyst pellet B is 1 mm in diameter.
(a) At the mid point in each catalyst (i.e. ![]() = 0.5) what is the midpoint concentration (CA @
= 0.5) what is the midpoint concentration (CA @ ![]() = 0.5) in catalyst A to the mid point concentration in catalyst B?
= 0.5) in catalyst A to the mid point concentration in catalyst B? ![]()
(b) What is the size of catalyst A?
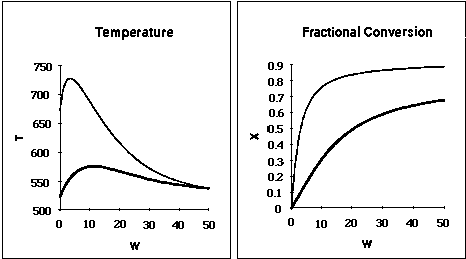
| T0 (K) | Tmax (K) | W(Tmax)* (kg) | Xfinal |
| 525 | 575 | 11.25 | 0.67 |
| 675 | 725 | 2.5 | 0.89 |
is carreid out on a zeolite catalyst. The following data was obtained in a differential reactor.
| Run | -rA mol/kg cat-min | PA (atm) | PB (atm) | PC (atm) | |
| 1 | 0 | 0 | 1 | 5 | |
| 2 | 0 | 1 | 0 | 0 | |
| 3 | 1.19 | 10 | 10 | 10 | |
| 4 | 0.8 | 1 | 1 | 1 | |
| 5 | 0.1 | 0.1 | 0.1 | 0.1 | |
| 6 | 0.59 | 1 | 5 | 0 | |
| 7 | 0.59 | 5 | 1 | 0 | |
| 8 | 0.8 | 1 | 1 | 0 | |
| 9 | 0.045 | 10 | 0.1 | 0 | |
| 10 | 0.045 | 0.1 | 10 | 0 |
is carried out adiabatically in a 100 dm3 PFR> Species A is fed to the reactor at a rate of 20 moles/min. The concentration of pure A is 3 mol/dm3. An inert stream is also fed to the reactor. The concentration of pure inert is also 3.0 mol/dm3. The entering temperature is 400K.
| FA0 = 20 moles/min CA0 = 3 moles/dm3 CI0 = 3 moles/dm3 | 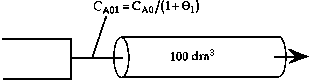
|
(b) First write out a POLYMATH program
(c) What is the ratio of the entering molar flow rate of inerts to enter molar flow rate of A, i.e. (![]() I = FI0/FA0) at which the conversion is a maximum? What is the maximum conversion? Hint (0.5 <
I = FI0/FA0) at which the conversion is a maximum? What is the maximum conversion? Hint (0.5 < ![]() I < 1.5)
I < 1.5)
Additional Information:
k = 0.0003 dm3/mol/min at 300K
E = 12,000 cal/mol/K
CpA = 10 cal/mol/K (species A)
CpB = 20 cal/mol/K (species B)
CpI = 40 cal/mol/K (Inert)
![]() HR = 10,000 cal/mol/K
HR = 10,000 cal/mol/K
is carried out in a moving bed reactor in which there is significant pressure drop. Pure A enters the reactor at a concentration of 0.1 mol/dm3 and a flow rate of 2 mol/min. The entering pressure is 10 atm and the pressure at the exit is 4.472 atm. The catalyst decay is zero order. The catalyst loading rate is 10 kg/min and the catalyst bed contains 40 kg of catalyst. What is the conversion at the exit of the reactor?
Additional information:
Specific reaction rate kR = 20.0 dm6/(mol-kg cat-min)
Decay Constant kD = 0.25/min
Back to the exam list