Report of the Medical Team of the Federal University of Rio de Janeiro on Accusations Contained in Patrick Tierney's Darkness in El Dorado 1*
| |
Dr. Maria Stella de Castro Lobo: Physician and Chief of Staff, Collective Health
Service of the Clementino Fraga Filho University Hospital, Federal University of
Rio de Janeiro (UFRJ), specialist in Epidemiology and Public Health. 2**
Dr. Karis Maria Pinho Rodrigues: Physician, Service for Infectious and Parasitic
Diseases of the Clementino Fraga Filho University Hospital , UFRJ, specialist in
Infectious Diseases. 3***
Dr. Diana Maul de Carvalho: Faculty member of the School of Medicine,
Department of Preventive Medicine, UFRJ.
Dr. Fernando Sergio Viana Martins: Faculty member of the School of Medicine,
Department of Preventive Medicine, UFRJ. |
The following report, written at the request of Dr. Bruce Albert, 4 anthropologist, and with his assistance, seeks to address various questions raised by the accusations contained in chapter 5 ("Outbreak") of the book by Patrick Tierney, Darkness in El Dorado, against the research team of the geneticist James Neel and the anthropologist Napoleon Chagnon. Tierney charges that they conducted experiments in 1968 using an inappropriate vaccine on the Yanomami Indians of the Orinoco River in Venezuela, which provoked or aggravated a measles epidemic. Therefore, we will examine and discuss various types of information, principally biomedical, in an effort to contribute to and illuminate, in the most objective manner possible, the terms of the polemic raised by the book, based on galley proofs of the chapter dated October 10, 2000, to which we had access.
1. Measles
Measles is a viral disease that is always considered severe. The first symptoms occur after 10 to 12 days of incubation, which represent the time between exposure to droplets of saliva from an infected individual and the onset of the illness.
The clinical profile begins with fever, malaise, conjunctivitis, coryza (runny nose), and tracheobronchitis (coughing), symptoms that comprise what is called the prodromal or precursory period, which lasts 2 to 4 days. Body temperature can rise as high as 40.6°C (105°F). Koplik's spots, the typical eruption (an enanthem present on mucous membranes) of measles that appears as punctate blue-white spots on the bright red background of the buccal mucosa, break out 1 to 2 days before the rash begins and lasts until 2 days after its appearance. The rash (or exanthem) corresponds to a morbiliform eruption, which usually appears 14 days after exposure and spreads from the head (along the hairline) to the trunk and extremities during a period of 3 days. During the next 3 or 4 days, the rash fades and the skin peels.
Measles is a highly contagious disease that affects almost all non-immunized individuals if exposed to the virus. Transmission occurs from person to person by means of respiratory droplets, but it can also occur through aerosol particles. The period of greatest transmissibility starts 2 to 4 days preceding the appearance of the rash and lasts until 4 days after it erupts. Therefore, after the index case (the first to occur), the appearance of secondary cases can occur on the average of 14 or 15 days (from 7 to 18) after exposure. There is no description of the disease being transmitted from asymptomatic individuals. The measles virus is quickly inactivated by heat, light, acidic pH, ether, and trypsin. It survives only a short time when exposed to air or on objects and surfaces (< 2 hours). Although susceptible monkeys can be infected, no animal reservoir exists for the measles virus. 5
The most frequent complications associated with measles are, in order of frequency: medium otitis, pneumonia, diarrhea, post-infection encephalitis, subacute sclerotic panencephalitis, and death. The lethality attributed to the disease is 1 to 3 cases/1000 in the U.S. 6 However, in developing countries, these rates can reach levels of 5 to 15%. Of these deaths, 60% result from secondary infections of bacterial pneumonia attendant upon measles. 7
2. The impact of measles on immunocompromised individuals and non-immune populations
In immunocompromised 8 individuals (carriers of congenital disease, HIV/AIDS, leukemia, lymphoma, generalized malignant disease, those using radiation therapy or medications that depress the immune system), measles can be prolonged, severe, and frequently fatal. In these individuals, the disease can occur without the presence of rash.
The effects of measles on non-immune indigenous peoples, such as the Yanomami, can also be extremely lethal. However, it is important to emphasize that individuals who are non-immune, due to a long period of isolation and the lack of previous vaccination, must not be considered immunocompromised. The seriousness of the cases and the high mortality rate found among isolated indigenous populations are basically due to the great concentration of susceptible individuals, conferring an explosive character to the epidemic when the virus arrives. 9
In the 1960s, a debate unfolded concerning the reason for the high rates of lethality occurring during outbreaks or epidemics of infectious diseases, such as measles, among indigenous peoples. According to James Neel, sociocultural aspects (lack of food storage and health care assistance) predominated over those strictly biological as the factors responsible for the alarming rates of lethality encountered.
Although a greater genetic homogeneity may predispose a group to a weaker capacity to respond, the immunological response to measles (level of antibodies after the disease or after vaccination) of recently contacted groups does not differ from that found in industrialized societies. 10 Neel expounded his thesis on this question in a scientific work published in 1970, based on research conducted during the measles epidemic of 1968 among the Yanomami of the Orinoco. Various other studies during that decade also demonstrated that health care at the time of the epidemic is the essential condition for reducing mortality. 11
3. Immunization Against Measles
Two forms of conferring immunity against measles exist. One form, called passive, is obtained by an injection of immunoglobulin. The disadvantage of this form is that it confers immunity for only 3 to 4 weeks. It is indicated in special situations as a means of avoiding the disease, such as after the exposure of children less than one year old, pregnant women, immunocompromised patients, and other individuals exposed to the disease who have some contraindication against the use of live vaccines (for example, an allergy to the components of the vaccine). It must be administered within 6 days of exposure.
The other form of immunization (called active) is through the use of a vaccine, which corresponds to the inoculation of an attenuated (i.e., weakened) live virus or a dead virus 12 in order to stimulate the immune system to produce antibodies that will keep the individual protected in case of exposure to the "wild virus" (the cause of the disease, properly speaking).
3.1) Edmonston B Vaccine
The measles virus was isolated for the first time in 1954 by Enders and Peebles; the strain was called Edmonston after the name of the child in whom the isolation occurred. After successive multiplications of this virus in different cellular types in order to attenuate it, the Edmonston B vaccine was produced, becoming licensed for use in the U.S. in 1963.
As time went on, Edmonston B was gradually substituted with other vaccines that were more attenuated, produced from the same strain of Edmonston, which had the advantage of inducing less intense vaccine reactions or side effects. These vaccines were the Schwartz vaccine, approved in 1965, and the Moraten, approved in 1968 and used at present in the U.S. It was not until 1975 that the Edmonston B vaccine was withdrawn from circulation in the U.S. In 1968 and 1969, for instance, more than 2 million doses of Edmonston B were still being administered in the U.S. 13
The immunity produced by the live virus vaccine is permanent and is similar to that produced by the disease. It must be measured through the amount of specific antibodies in the blood serum. The levels of antibodies are typically lower after the vaccination than those related to the disease. The levels of antibodies also diminish over time and can become undetectable. However, this does not mean that the vaccine no longer offers protection.
3.2) Side Effects
Although the use of Edmonston B is associated with a high frequency of fever (39.4°C, or 102.9°F) (in 20-40% of cases) and rash (in 50%) occurring 5 to 12 days after the vaccination, there are no reports of serious complications. In general, individuals remain well, with a less severe profile than with the measles disease, and there are no reports of deaths resulting from this reaction among immunocompetent individuals.
The use of immunoglobulinIG or MIG (high-titer measles immune globulin, no longer available in the U.S.)along with Edmonston B reduces the occurrence of these symptoms by about 50%. The Schwartz and Moraten vaccines do not require the associated use of immunoglobulin.
It should be noted that, although the vaccine virus was isolated in the conjunctiva and respiratory tissues of a single individual with leukemia, who died as a result of pneumonia one year after vaccination, the transmission of the vaccine virus from person to person has never been documented (see Redd et al., cited above in note 4).
In the case of non-immune populations in general, and indigenous peoples in particular, various studies have shown that the greater incidence and intensity of post-vaccine reactions encountered among vaccinated individuals were characterized by fever, prostration, and rash, simulating the profile of light to moderate cases of measles. Some of these studies, despite the difficulties of comparing groups with similar controls, recommend the use of vaccines containing more attenuated forms of the virus when access is difficult for health teams to attend to adverse effects. 14
3.3) Conditions for Administering Measles Vaccines (with attenuated virus)
It is usually recommended that measles vaccines not be administered to individuals with fever, since the concomitance of the illnesses can diminish the rate of serum conversion. 15 However, vaccination should not be delayed in the case of moderate respiratory infections, since studies have shown no difference in the serum conversion under such conditions. Furthermore, in studies that compared vaccination between malnourished and well nourished children, the rates of serum conversion were identical. 16
The use of these vaccines is not recommended in immunocompromised individuals. However, specifically with regard to HIV infection, this recommendation has been changed, since the number of infected children has increased and the risk of severe disease in these children is very high. Studies in Africa have shown few side effects, such that now in the U.S. it is also recommended that children without signs of advanced immunodeficiency be vaccinated. 17
The use of vaccines after exposure to the measles virus can be effective by offering some protection against the onset of illness if it is administered within 72 hours after exposure. This protection is made possible because the time needed to produce antibodies is less in the case of the vaccine, since it is administered outside the alimentary tract (parentherally). This is the preferred option when treating individuals over 12 months old. There is no contraindication against the use of the vaccine after the 72-hour period. However, if administered late, the vaccine may not be effective in preventing the illness or reducing the symptoms, and the individual may come to present the full-blown disease. It should be noted that this does not represent a failure of the vaccine or complications with it (see note 4 above). Finally, in the midst of an epidemic, the recommended practice is mass vaccination to erect a barrier against the spread of the disease, even if there are concurrent diseases or malnutrition at the time of the vaccination.
4. Ethics and Biomedical Research
At the present, the ethics guiding research on health issues in Brazil are based on four fundamental points, all of which are detailed in "Policies and Regulations for Research Involving Human Subjects," Resolution 196/96 of the National Health Council:
A) Qualifications of the team of researchers and of the project;
B) Evaluation of the cost/benefit ratio;
C) Informed consent;
D) Prior evaluation of the ethical aspects of the research project by an ethics committee, composed of researchers and community representatives.
In the early 1960s, before the research team of James Neel and Napoleon Chagnon conducted their research among the Yanomami, the Declaration of Helsinki I had been adopted by the 18th World Medical Assembly (1964), which dealt with the principles inherent in the first three issues (above). Informed consent had been a prominent recommendation ever since the International Tribunal of Nuremberg of 1947 established the Nuremberg Code, which states:
1. The voluntary consent of the human subject is absolutely essential. This means that the person involved should have legal capacity to give consent; should be so situated as to be able to exercise free power of choice, without the intervention of any element of force, fraud, deceit, duress, over-reaching, or other ulterior form of constraint or coercion; and should have sufficient knowledge and comprehension of the elements of the subject matter involved as to enable him to make an understanding and enlightened decision. This latter element requires that before the acceptance of an affirmative decision by the experimental subject there should be made known to him the nature, duration, and purpose of the experiment; the method and means by which it is to be conducted; all inconveniences and hazards reasonably to be expected; and the effects upon his health or person which may possibly come from his participation in the experiment. The duty and responsibility for ascertaining the quality of the consent rests upon each individual who initiates, directs or engages in the experiment. It is a personal duty and responsibility which may not be delegated to another with impunity. 18
5. Discussion
In light of this framework of ethical guidelines for biomedical research in vigor during the 1960s, it is possible to make some comments on the procedures of the research project carried out by the team of Neel and Chagnon in relation to the accusations made in chapter 5 of Tierney's book.
A) Concerning the Qualifications of the Researcher and the Research Project
James Neel was an academic scholar of international renown, both then and now, with recognized competence to plan, execute, and disseminate the results of a research project. However, the research protocol and the principle objective of his investigation among the Yanomami of Venezuela in 1968 were not clearly explained in the article by Neel et al., published in the American Journal of Epidemiology in 1970 (see note 7 above), which describes the vaccination and the measles epidemic.
In this article, the authors refer to the disruption of their research protocol due to the necessity of emergency care for the Yanomami, given the simultaneous outbreak of the epidemic. They also allude to the initial research objective, which was to have been a comparison of the response to the measles vaccine between a "truly primitive" population and a control group of "civilized" individuals. According to Ryle Ward, professor at the University of Oxford and former collaborator of Neel, the latter intended to vaccinate 2,000 Yanomami and return a few years later to conduct a new serological study in order to measure their immunological response to the vaccine.
However, Tierney's book suggests that the objective of the research was to conduct an experiment inspired by eugenics, which would detect differences in genetic susceptibility and analyze natural selection among the Yanomami by means of an epidemic introduced through vaccination ("to resolve the great genetic question of natural selection"). Such an intention, which would have been absolutely unethical, seems to us in this case to be highly improbable. In the first place, there is no proof whatsoever that the vaccine virus is transmissible and, thus, capable of generating an epidemic. In the second place, Neel himself, in an article published in 1977, asserts that the number of individuals needed to detect a process of natural selection would be enormous, and that, in methodological terms, it would be practically impossible to find evidence of differences in small populations (as in the Yanomami case), even if they existed. 20 Moreover, Neel's hypothesis was that sociocultural factors predominated over genetic ones as those responsible for the excessive mortality caused by recently introduced infectious diseases among isolated indigenous peoples (see the article by Neel et al., 1970, cited above).
In conclusion, the principle research objectives of Neel associated with the Yanomami vaccination in 1968 and the protocol of this research can be evaluated only after obtaining the original research statement presented to the AEC (the U.S. Atomic Energy Commission, which financed the research) and, preferably, by an ethics committee composed of independent specialists (as required by current standards).
B) Concerning the Cost/Benefit Analysis of the Vaccination
For this criterion, the advantages of a procedure for the population in question must be weighed against the possible complications, known and unknown, inherent in the same procedure. In this context, conducting the vaccination as a prevention against measles appears to us to have been a beneficial measure, since the Yanomami population found itself both vulnerable, given the intensification of interethnic contact, and biologically susceptible, given the high percentage of those lacking antibodies against measles, as determined from blood samples collected in 1966-67 (cited in Neel et al., 1970).
However, it is worth noting that during the same decade, and in earlier ones, other studies were conducted among the Yanomami that had no clear benefit for this population, 21 including, among others, research on thyroid metabolism, which was also supported by an atomic energy commission, this time the French one (CEA). 22
B.1) On the Choice of the Edmonston B Vaccine. A few considerations can be highlighted regarding the choice of the Edmonston B vaccine by Neel. The first is that he used this vaccine after asking for technical information from the Centers for Disease Control (CDC) in Atlanta, according to the findings of Susan Lindee (Department of History and Sociology, the University of Pennsylvania), who examined Neel's personal documents. 23
Ever since the release of Edmonston B for measles in 1963, studies revealed a greater frequency of reactions to the vaccine among isolated groups (see Brody et al., 1964), leading to the choice of vaccines with more attenuated forms of the virus. Although the use of Edmonston B was still valid when the vaccination took place in 1968, having been used on around one million American children in the same year, the Schwartz vaccine, being more attenuated, presented the theoretical possibility of a vaccine that would cause fewer complications while offering the same efficacy.
On the other hand, some clinical comparative trials were still being conducted in 1968 on Edmonston B and Schwartz to compare the frequency of reactions to the two vaccines, 24 which suggests that, at the time, such theories were still only hypotheses under experimentation. The work cited above went so far as to compare the two vaccinated groups with a third group that received a placebo (distilled water), a procedure that nowadays would be considered ethically inadmissible.
Once again, we do not have access to documents that could answer clearly why Neel chose this or that vaccine for vaccinating the Yanomami in 1968 in Venezuela (and in 1967 in Brazil, when 1000 doses were sent to local evangelical missionaries for vaccinating the Yanomami in Toototobi, Surucucus, and Mucajaí). In any case, it should be mentioned that the monitoring of the ethical aspects of medical research among indigenous peoples was, in the 1960s, quite distant from current conceptions, despite the rules listed in the Nuremberg Code (1947) and the Declaration of Helsinki (1964).
Tierney's book presents an argument that the Edmonston B vaccine used in Neel's research, besides having been phased out, carried an elevated risk of serious complications, even lethal ones, rendering it absolutely contraindicated in the Yanomami situation. 25 As stated above in item 3.2, although the Edmonston B vaccine can produce a greater percentage of vaccine reactions, mainly among isolated groups for whom the measles disease is also more serious, these reactions take the form of fever and rash, without the risk of death.
It is worth noting that the information presented earlier in the book predisposes the non-specialist reader to believe, despite the lack of documentary support, the worst about the researcher's intentions, who supposedly chose this vaccine in order to provoke a simulation of a measles epidemic. An example of this is the exaggerated attention given in the text to the composition of the vaccine with live virus, in contrast to that with the dead virus, when, to the contrary, it is the latter that carries a much greater risk if followed by exposure to the disease virus (which is the reason why it was no longer recommended in 1967). 26
Finally, it is also worth pointing out the conceptual confusion in Tierney's book concerning the immunity of the Yanomami. During the time of the Yanomami vaccination in 1968, much debate took place over the preponderant factor behind the high lethality of measles among such populations, but this does not justify characterizing or comparing the Yanomami with individuals who are immunocompromised 27 (see clinical examples cited above in item 2 and 3.3). An example of this confusion is the clinical profile described in chapter 5, which likens measles among the Indians to measles among leukemia patients, which amounts to a falsehood. 28 Along similar lines, Tierney attempts to demonstrate that the Yanomami are immunocompromised by means of anthropometric measurements (BMI, the Body Mass Index, which divides the weight in kilograms by the square of the height in meters), leading him to characterize this population as systematically malnourished. Although the BMI may be considered an excellent indicator of the state of health of a population, it is necessary to exercise considerable caution when comparing specific biotypes with international standards of normality. 29
B.2) On the Selective Use of Immunoglobulin (MIG). As an additional point of discussion, although the concomitant use of immunoglobulin with Edmonston B was frequently recommended in order to reduce vaccine reactions, many studies were conducted without immunoglobulin to compare these reactions in the absence of any intervening factor. 30 In the case of the vaccination conducted by Neel in 1968, little is known about whether he had a deliberate interest in comparing groups vaccinated with and without measles immune globulin (MIG).
It is known that the vaccination was conducted without immunoglobulin (MIG) in Venezuela (near the Ocamo Mission), as well as in at least one of three regions in Brazil (Surucucus, Toototobi, and Mucajaí), where 1,000 doses of Edmonston B were sent (see Neel et al., 1970:423). In the Venezuelan case, the only group that did not receive immunoglobulin (MIG) was the first to be vaccinated, so one might speculate that an initial protocol of comparing the reactions to the vaccine with or without MIG, in other villages as well, was perhaps aborted given the epidemic emergency.
In any case, evidence that a comparison was indeed made between people who were vaccinated with and without immunoglobulin (MIG) can be found in the 1970 article of Neel et al. The editors of the American Journal of Epidemiology, by accepting it for publication, apparently did not consider the issue to be a serious ethical problem at that time.
Given this fact, the hypothesis becomes plausible that Neel may have actually intended to conduct this comparison in planning the vaccination. Such a procedure, which today we would certainly evaluate as ethically inappropriate, was, however, apparently compatible with the practices of biomedical research and the editorial policies of the 1960s (but not with the rules of the 1947 Nuremberg Code and the 1964 Declaration of Helsinki).
Finally, it remains for us to comment on one more question in the case, one that was not even broached by Tierney's book, which concerns the conclusion of the article by Neel et al. (1970). In the midst of an epidemic, when it is impossible to differentiate accurately between cases of measles and vaccine reactions, it would hardly be scientific to say, based on this experience, that the reactions were more frequent among those vaccinated without an injection of immunoglobulin (MIG). 31 But again, this shows that the methodological standards of scientific journals in the 1960s were very different than those currently in effect.
B.3) On Withholding Assistance and Selective Vaccination. More than once, Tierney claims that Neel and his team did not adequately treat Yanomami who had symptoms of measles, by preventing the use of antibiotics, avoiding requests for more medical personnel, and even failing to vaccinate villages through which he passed. However, Tierney offers no solid documentation in chapter 5 that would support such statements. For example, to support the charge that no vaccination was carried out in Platanal, he refers to the article by Neel et al. (1970), in which there is no mention whatsoever of such a fact. Besides this, much of his information is based on his interpretations, inevitably open to diverse appraisals, of fragments of interviews and especially film soundtracks.
From what is actually possible to assess in this case, we know that Neel tried to create a barrier against the epidemic by means of the vaccination, which, given the speed with which the disease spread, was not sufficient to reduce the mortality that occurred. Furthermore, it is worth underscoring that it is not always easy to distribute the tasks and human resources in the dramatic circumstances of an epidemic in an indigenous area. Also, it would have been practically impossible to alter the Yanomami's behavior (given their mobility) and impose a quarantine on villages that were isolated or little contacted, a measure that Tierney suggests.
On the other hand, given that Neel was already aware of the risk of the epidemic during his preparations for the trip (having received information on the epidemic in Brazil in the border regions with Venezuela in late 1967), some additional precautions could have been included or foreseen in his work plans. Some precautions that would have reduced the difficulties encountered in the field include the training of those administering vaccinations, information on complications and treatments, provisions of medications and antibiotics, an itinerary and schedule of villages to visit, etc.
If, in the final analysis, it does not seem to us that there is conclusive documentation to substantiate the accusation that Neel's research team refused to vaccinate or treat any case of vaccine reactions or complications from the measles disease, the motives for not vaccinating all the individuals in the villages visited by the team remain to be clarified. On the one hand, the accusation raises questions about whether this could be evidence of medical neglect or even of the constitution of a control group as part of an experiment. On the other hand, one must also consider that the traditional mobility of the Yanomami in isolated areas, particularly in the context of a panic in the midst of an epidemic, in addition to the lack of censuses and records for identifying individuals, can explain why some villages or parts of villages were left out during the vaccinations.
However, this last explanation may seem less plausible in the failure to immunize 36 people from Iyewei theri at the Ocamo Mission, where only 31 residents were vaccinated. Since this was a village situated near a missionary station, founded in 1957, these Indians would have been less mobile, and the mission must have had a census list. Therefore, they would have been more accessible, but they were not all vaccinated during the three weeks following the date when the immunization in Ocamo began. 32 In this case, there is a lacuna in the information about the reasons for the absence of those Indians at the mission during the vaccination period.
C) Concerning Informed Consent
Resolution 196/96 of the National Health Council in Brazil, requires that, in culturally differentiated populations, including indigenous ones, prior assent be granted by the community through its own leaders, which does not, however, preclude efforts to obtain individual consent. Besides these official norms, the Yanomami and many other indigenous groups are today capable of expressing knowledgeable opinions about conducting biomedical experimentation; there are even forums for indigenous representation in the 34 Special Indigenous Health Districts established by the National Health Foundation (FUNASA, in the Ministry of Health) in various states in the country. In this context, the former practice of exchanging gifts for blood (used by the team of Neel and Chagnon with the Yanomami and other indigenous groups), or any other similar procedure that substitutes as a distorted form of "informed consent" from indigenous populations, is nowadays totally prohibited by national legislation, as well as by indigenous communities and organizations in Brazil and worldwide.
If we consider authorization for biomedical research among indigenous peoples from the perspective of today's standards in Brazil, we find that Regulation #01/95 requires that "every researcher, including citizens and foreigners, who wishes to enter into indigenous territory to carry out scientific research projects must submit a request to the president of FUNAI" (Fundaçăo Nacional do Índio, the National Indian Bureau). Moreover, Resolution 196/96 made the National Commission on Research Ethics (CONEP) of the National Health Council responsible for approving and monitoring research projects in special thematic areas, among these being research on indigenous populations, projects directed from abroad or with foreign participation, and research that involves sending biological samples to foreign countries.
There still remains, in its context, the question of authorization from the Venezuelan health authorities for Neel's team to conduct research and vaccinations. Again, certain doubts exist about the researcher's posture in this respect.
On the one hand, Tierney's book presents a statement from Dr. A. Bitencourt, who, at the time, was Director of the Department of Vaccinations in the Venezuelan Ministry of Health, in which she asserts that she did not receive any request for authorization for the research. In addition, journalists who carried out investigations in Brazil, where Neel and his collaborators collected blood for genetic studies during at least six expeditions among indigenous groups (including the Yanomami in 1967, with Napoleon Chagnon), found evidence that some of these expeditions were conducted without official authorizations or else through authorizations obtained by local colleagues. 33
On the other hand, a document exists that demonstrates Neel's intention of obtaining official authorization, at least for the vaccination program in 1968 in Venezuela, being a letter written to Miguel Layrisse, his colleague at the Venezuelan Institute of Scientific Investigations (IVIC), on December 11, 1967. 34
Finally, whether the Venezuelan authorities actually gave their consent for the vaccination program in 1968, or for a research protocol associated with this program, is a question about which, once again, we do not have conclusive information at this time.
6. Alternative hypothesis on the origin of the measles epidemic in the Orinoco region (1968)
The argument in chapter 5 of Tierney's book leads the uninformed reader to the conclusion that the Edmonston B vaccine was supposedly chosen for its ability to start a measles epidemic. As we have already stated, there is no description in the medical literature of person-to-person transmission of the vaccine measles virus, nor even of transmission via objects changing hands, since the virus is rapidly inactivated outside the host's body. 35
To support his hypothesis, the author starts with the assumption that the first case of measles diagnosed by Neel was not really a case of the disease, and therefore the explanation for the arrival of the measles virus in the community would have been the Edmonston B vaccine itself. Given the absence of rash in the first patient they examined, mentioned by Neel in the article describing the epidemic (Neel et al., 1970), such a diagnosis of measles was, in fact, uncertain (sometimes it is difficult to discern a rash in non-white populations). On the other hand, it is possible that this diagnosis was determined retrospectively, since it was the first case of fever recorded by the team before the epidemic was confirmed. The diagnosis could have been substantiated only if blood had been collected from the patient to measure the amount of antibodies before being vaccinated. In any event, even if this individual was not the first case of the disease, this does not support the hypothesis that the epidemic resulted from the vaccine.
In the face of these speculations and doubts, the best way of approaching the enigma of the measles epidemic in the Orinoco region in early 1968 and understanding the epidemiology of its origin and dissemination would be to determine the chain of transmission of the disease. We have sought to do this through a chronological ordering of the facts tangled up in chapter 5 of Tierney's book, the article by Neel et al. (1970), and information conveyed in other documents, including a letter from the researcher (see chart and map below).
The measles epidemic among the Yanomami in Brazil in 1967, cited in the article by Neel et al. (1970), has been confirmed by various sources. The daughter of a missionary from the New Tribes Mission (NTM), came from Manaus to Toototobi (a tributary of the upper Rio Demini), bearing the disease, which infected the Yanomami in that region and launched an epidemic that lasted from September to November of 1967. Despite the permanent health assistance dispensed by the missionaries, the mortality rate was 15%, similar to that described in the literature for developing countries. 36 A measles epidemic was also reported among the Brazilian Yanomami on the Rio Apiaú (Rio Mucajaí basin), Rio Ajarani, and the Repartimento Stream (Rio Catrimani basin) in April and May of 1967. 37
Chronology of Events Cited
| DATE |
EVENT |
| Dec. 11, 1967* |
 Neel indicates he was already informed of the measles epidemic Neel indicates he was already informed of the measles epidemic
among Yanomami of the upper Demini (Toototobi, Brazil) and possibly
on the upper Orinoco (Venezuela) by missionaries of the New Tribes
Mission |
| Jan. 22, 1968 |
 Arrival of Neel and Chagnon at the Ocamo Mission (Venezuela) Arrival of Neel and Chagnon at the Ocamo Mission (Venezuela)
 Account of 5 deaths with respiratory symptoms during the 2 Account of 5 deaths with respiratory symptoms during the 2
preceding months near confluence of Mavaca and Orinoco rivers |
Jan. 22-25,
1968** |
 First case of measles diagnosed (without rash) on Jan. 22; patient First case of measles diagnosed (without rash) on Jan. 22; patient
vaccinated
 Beginning of the anti-measles vaccination at Ocamo (in Iyewei Beginning of the anti-measles vaccination at Ocamo (in Iyewei
theri, 31 residents vaccinated, plus visitors from other villages,
totalling 40 people vaccinated)
 3 people from Iyewei theri move to Shubariwa theri, where high 3 people from Iyewei theri move to Shubariwa theri, where high
mortality is later reported, with the exception of cases treated by a
nurse |
| Jan. 29, 1968 |
 First description of rash among individuals vaccinated on Jan. 23 First description of rash among individuals vaccinated on Jan. 23 |
| Feb. 1, 1968 |
 Death of 2 children who had been vaccinated on Jan. 24 Death of 2 children who had been vaccinated on Jan. 24 |
| Feb. 2, 1968 |
 36 ill patients treated at Ocamo Mission (among total of 40 36 ill patients treated at Ocamo Mission (among total of 40
people vaccinated)
 Seriously ill child, daughter of local administrator, brought to Seriously ill child, daughter of local administrator, brought to
Mavaca Mission |
| Feb. 4, 1968 |
 First notation about measles epidemic in medical records at Ocamo First notation about measles epidemic in medical records at Ocamo
 Visit by two employees from Platanal Mission to Ocamo Visit by two employees from Platanal Mission to Ocamo |
| Feb. 4-8, 1968 |
 Neel's article describes 2 more symptomatic individuals, Brazilian Neel's article describes 2 more symptomatic individuals, Brazilian
Amazonian peasants (caboclos)
 Vaccination of people in Witokai theri, who subsequently moved Vaccination of people in Witokai theri, who subsequently moved
to Mavaca |
| Feb. 15, 1968 |
 Death of child with rash brought earlier (Feb. 2) to Mavaca from Death of child with rash brought earlier (Feb. 2) to Mavaca from
Ocamo
 Beginning of vaccination at Mavaca Beginning of vaccination at Mavaca |
| Feb. 17, 1968 |
 Vaccination of 65 Yanomami in Coshirowa theri Vaccination of 65 Yanomami in Coshirowa theri
 17 cases of measles among the 36 people at Iyewei theri who 17 cases of measles among the 36 people at Iyewei theri who
were not vaccinated at Ocamo (symptoms began on Feb. 16) |
| Feb. 18, 1968 |
 Filming of the epidemic begins at Mavaca (typical morbiliforme Filming of the epidemic begins at Mavaca (typical morbiliforme
rash)
 At Mavaca, illness reported in Bisaasi theri (where vaccination At Mavaca, illness reported in Bisaasi theri (where vaccination
took place Feb. 15) among individuals who came from Witokai theri
Feb. 4-8
 Rerebawa, Chagnon's assistant from Karohi theri, displays fever Rerebawa, Chagnon's assistant from Karohi theri, displays fever
and Koplik's spots
 Request for doctors and penicillin made through radio contact Request for doctors and penicillin made through radio contact
 Neel's team departs for Platanal (to vaccinate people in Mahekoto Neel's team departs for Platanal (to vaccinate people in Mahekoto
theri, Hasupuwei theri, and Patanowa theri), leaving a nurse at
Mavaca to continue treatments |
| Feb. 19, 1968 |
 Arrival of Neel's team at Platanal Arrival of Neel's team at Platanal
 Tierney's accusation claims team did not vaccinate at Mahekoto Tierney's accusation claims team did not vaccinate at Mahekoto
theri, citing article of Neel et al. 1970 (?)
 Photo of 2 employees who were healthy upon arrival at Ocamo Photo of 2 employees who were healthy upon arrival at Ocamo
on Feb. 4; according to Neel, they later became symptomatic
 Report made (after the fact) of illness and death of 25% of Report made (after the fact) of illness and death of 25% of
Mahekoto theri residents (no date specified) |
| Feb. 22, 1968 |
 Reference to trip through Patanowa theri Reference to trip through Patanowa theri |
| Feb. 26-28, 1968 |
 Fever profile among 43 of those vaccinated at Coshirowa theri Fever profile among 43 of those vaccinated at Coshirowa theri |
* Letter from J. Neel to M. Layrisse, cited above in item C.
** The precise date of the beginning of the vaccination is not clear in either Tierney's text nor in the article by Neel et al. (1970).
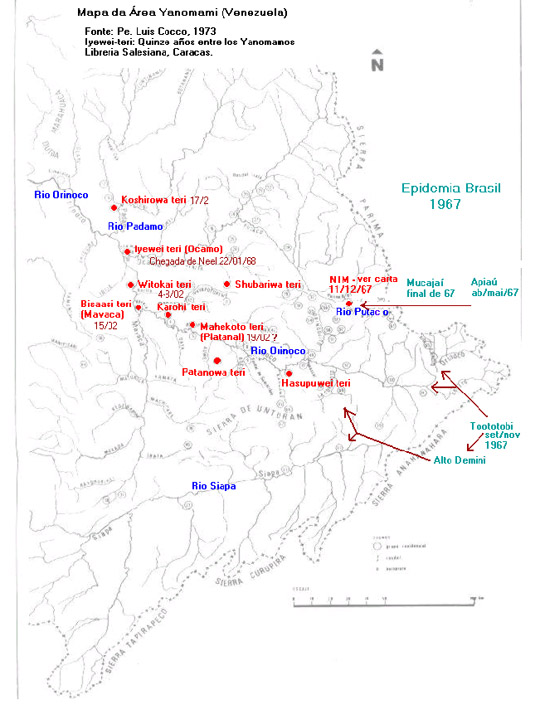
It should be noted that the Rio Toototobi is very close to the border with Venezuela and that the Yanomami of the region say that the epidemic reached the Yanomami of the Rio Siapa and the upper Orinoco. In fact, the Yanomami of the former Toototobi Mission used to maintain regular intercommunity relations with the groups on the upper Rio Demini (Parawau)—which, in turn, took part in an exchange network that extended to the communities of the upper Siapa˜as well as with groups at the Orinoco headwaters.
In addition, a letter from James Neel to Miguel Layrisse, dated December 11, 1967, also reports recent information from the NTM missionaries that "there is sickness amongst the Indians on the very high Orinoco, possibly due to measles." This probably refers to the region of the upper Putaco, a tributary of the Ocamo where a NTM station was located. This region is also close to the Brazilian border and the headwaters of the Parima and Mucajaí Rivers (the letter also mentions measles on the Rio Mucajaí).
All these data raise the possibility that the paths of propagation taken by the disease were through the network of relations among the Yanomami villages, starting in the region of Toototobi and/or Mucajaí in Brazil and heading in the direction of the upper Orinoco in Venezuela, either by a northern route (Rio Mucajaí—Rio Parima) or a southern route (Rio Toototobi—upper Demini—upper Siapa, and Rio Toototobi—Orinoco headwaters), or both. 38
Although they are hypothetical, these possible chains of transmission, whatever form they took, appear to be much closer to the truth as an explanation of the onset of the epidemic in the Orinoco than the theory presented in Tierney's book, which attributes the epidemic to the Edmonston B vaccine.
When Neel's team arrived at the Ocamo Mission, there were reports of a series of cases of severe respiratory diseases in the region during the two preceding months, which were diagnosed as bronchial pneumonia, but which, according to statements gathered by Tierney, curiously did not respond to antibiotic treatment. Although the article by Neel et al. (1970) states that the response to penicillin was satisfactory, this new information makes us question the diagnosis suggested (bronchial pneumonia) and infer that the disease had a viral etiology, perhaps from an epidemic of influenza or even measles. 39
Although the documentation is imprecise concerning the exact date of the beginning of the vaccination (in Neel et al. 1970:421, it appears that it was January 22, 1968, or shortly thereafter), some indications suggest the means by which the disease disseminated. If the Yanomami of the Ocamo Mission were vaccinated more than 72 hours after contact with the wild virus—the period after which vaccination is generally no longer effective for preventing or lessening the impact of measles—the presence of visiting Indians at the mission and their movement to other villages (for example, to Shubariwa theri) allows us to determine the velocity of the spread of the epidemic. If the rash appears, on the average, 14 days after the contagion, and the first cases of the rash are dated approximately January 29, 1968, 40 these Indians would have had contact with the disease on January 15, 1968. Therefore, exposure would have occurred one week before the arrival of Neel's team and the vaccine inoculation, which would explain why the patients were in such serious condition.
The report of the first deaths from bronchial pneumonia, a complication from measles, on February 1, 1968, is also more compatible with a possible contagion in the middle of January. On the same day, a sick child was transferred to Mavaca, bringing the wild virus 15 days before the vaccination took place at the mission. Shortly afterwards (February 4, 1968), in the midst of the documented epidemic, two employees from Platanal Mission came to Ocamo, who also may have carried the virus back with them. During this period, Indians vaccinated at Witokai theri, the village closest to the Ocamo Mission, also moved to Mavaca, where they soon presented symptoms of the disease.
Accordingly, Neel appears to have arrived at Mavaca too late. On the date of his arrival (February 18, 1968), the appearance of Koplik's spots on Chagnon's guide also leads us to believe that he was infected in Witokai theri (or in the village where he resided, Karohi theri), 10 to 14 days earlier, where the measles virus must have already circulated. The date of infection would have also been in the beginning of February in the case of the 17 individuals in Iyewei theri who became ill, being some of those who had not been vaccinated at Ocamo at the beginning of the expedition.
Once again, the arrival of the health team in Platanal appears to have been too late (consider the visit of the employees to Ocamo), explaining the subsequent epidemic and the high rate of mortality observed in Mahokoto theri. In the case of the residents of Coshirowa theri, who presented the first symptoms 10 to 12 days after the vaccination (see Neel et al., 1970), it becomes even more difficult to differentiate the vaccine reaction, properly speaking, from the profile of the measles disease that was lessened by the vaccination, this time administered soon enough (in this case, no deaths were reported). It should be noted that this village is situated to the north of Ocamo, that is, more distant from the circuit of the most intense contagion, which was to the southeast of the mission.
One possible conclusion from this reconstitution of the epidemiological chain would therefore be that, instead of having caused the epidemic, the vaccinations by Neel's team appears to have not been sufficient to prevent the mortality observed in some of the villages. In other words, the villages that were visited later than 72 hours after contact with the measles virus could not benefit from the intended protection, which explains the disastrous impact of the disease. In this case, the eruption of symptoms of measles shortly after the vaccination, which left such an impression on various observers cited by Tierney, can be explained by the appearance of the measles disease and not by an exaggerated reaction to the vaccine.
Finally, if these chronological facts make it possible to demonstrate some of the chains of transmission in the upper Orinoco, we presume that many other chains unknown to us must have also occurred in a complementary form, which makes it impossible to identify the illness at the Ocamo Mission as the index case. The observation and analysis of the map above also allows us to conclude that the arrival of the wild virus in Venezuela occurred in an east-to-west direction, while the movement of Neel's team was predominantly in the opposite direction, that is, against the epidemic's movement. Their path was justified by the greater probability of finding villages needing urgent medical assistance.
1. Conclusions: what can and cannot be concluded
Cause of the epidemic and its lethality
 |
The principle hypothesis of chapter 5 in Patrick Tierney's book—that a measles
epidemic was provoked by vaccinations as part of a eugenics experiment—are easily refuted, his arguments having little solidity and being highly inconsistent. |
 |
At the time when Neel's team conducted the vaccinations, other vaccines were available that provoked fewer reactions than the Edmonston B they chose. Nevertheless, the intention of using this vaccine cannot be considered technically incorrect. |
 |
The mortalities observed in 1968 on the Rio Orinoco were probably the result of a true measles epidemic that came from Brazil, with no relation to the vaccination (although concurrent with it); generally speaking, the vaccinations were not conducted soon enough to block the epidemic. It is worth emphasizing that putting up a barrier around an epidemic by means of vaccinations continues to be the recommended practice up to today. Since Neel was aware of the speed with which the disease spread and the difficulties encountered in this type of fieldwork, the only question is whether, given his prior knowledge of the epidemic in Brazil, better planning and training of the teams of vaccinators could have reduced the impact of the epidemic. |
Planning and conducting experiments
 |
Administering Edmonston B along with immunoglobulin (MIG) was the recommended procedure in those days to counteract vaccine reactions, although the use of the vaccine alone was not contraindicated. In fact, the use of the vaccine without immunoglobulin was compatible with the state of the art in research during the 1960s. However, clinically the use of associated immunoglobulin would be more indicated, considering the well-being of a population, such as the Yanomami, that was isolated and susceptible to the most intense side effects. The true reasons for which Neel opted not to use immunoglobulin (MIG) among 31 of those vaccinated at Iyewei theri (and in at least one of the three regions in Brazil) are not clear from the available documentation, although the intention of experimentation is plausible. |
 |
Although Neel mentioned in his article the disruption to the research protocol and methodology he initially projected (which he did not clearly explain), we stress that, in any case, the results of his comparison of individuals who were vaccinated with and without MIG are of dubious value, insofar as individuals with a vaccine reaction cannot be differentiated from those who already have measles, modified or not by the vaccine. |
Ethics in research among indigenous peoples, past and present
 |
A veritable dichotomy can be observed in the 1960s between the state of the art of ethics in biomedical research and the actual practices that were followed, especially among minorities. In this context, the experimentation that may have been conducted by Neel would have been counter to the ethical precepts, but would not have differed in this respect from many other research projects on human subjects carried out and published in renowned journals in that decade. |
 |
The positive aspect of the polemic raised by chapter 5 of Tierney's book, despite its serious documentary and conceptual failures and its lack of demonstrative rigor, is in the fact that it has made possible a more profound discussion reflecting upon the ethics of research among indigenous populations and minorities in general, not only in biomedical research, but also in other spheres, such as anthropological research, which, in the case under discussion, was strictly associated with biomedical research. |
 |
Finally, if the scientific community judges it necessary to probe deeper into the specific questions raised by the accusations in Tierney's book and discussed here in this report, we suggest the creation of an international committee, independent and multidisciplinary, which would proceed to analyze the case based on the protocol of the research project, documents from government institutions that were involved at the time, field notes from the team of James Neel and Napoleon Chagnon, and records of medical treatments administered in the field. |
Postscript (November 18, 2000)
As mentioned at the beginning of this report, our analysis of chapter 5 of Patrick Tierney's book was based on the page proofs, which circulated before its publication and which triggered a debate that was widely covered in the national and international press starting in September, 2000.
Confronted by the intense controversy that surged in academic and journalistic circles on the thesis presented in the first version of the chapter, the author ended up introducing some modifications in the version that was published in mid-November, 2000, a highly unusual editorial procedure. The majority of the alterations tend to soften the formulation of his previous accusations (for example, p. 61 on the MIG experimentation; p. 75 on Neel's lack of medical care for sick patients; p. 80 on the Edmonston B vaccine and the violation of medical ethics). Others deal with some of the statements he collected, generally with the same function (p. 61, where the interview with Dr. C. Bott is deleted; p. 80, where the interview with Dr. Papania is contextualized and the interview with J. Earle is deleted). However, the main modification consists in the complete restructuring of the chapter's conclusion (pp. 80-82). Below, we add some commentary on the book's new concluding section.
 |
Technical references of a biomedical character were amplified (notes 138, 140, 141), which, nevertheless, do not substantiate the hypotheses presented in the original text (of the page proofs). On the contrary, these additions make the author's argument on the vaccine origin of the epidemic even more ambiguous. Thus, the new text juxtaposes a categorical assertion by Dr. Samuel Katz on the impossibility of a measles vaccinal transmission alongside comments that, in an indirect manner, leave doubts about Katz's statement. |
 |
Further reinforcing this new ambiguity ("it is unclear whether the Edmonston B became transmissible or not"), Tierney ends up attributing the mortality encountered during the 1968 epidemic to the association of the vaccine reaction with malaria and bronchial pneumonia, further aggravated by the behavior of the Yanomami when they panicked and dispersed as the epidemics arrived. Claiming that it is simplistic to argue in terms of a "measles epidemic," he proposes the notion of a "measles vaccine-bronchial pneumonia-malaria epidemic." Although Tierney cited the occurrence of cases of bronchial pneumonia and malaria during the vaccination (pp. 62-63), there is no indication whatsoever in the text (nor in the article by Neel et al., 1970) of such a clinical profile and/or about serious complications due specifically to malaria (P. falciparum), such as cerebral malaria, which frequently lead to the deaths of Yanomami struck by this disease. Moreover, according to what the author himself reported on previous pages, measles among "virgin-soil" indigenous peoples is sufficiently serious to explain the high mortality encountered during the 1968 epidemic. Therefore, the most convincing hypothesis for the observed mortality is still the measles epidemic itself, which, recognizing its seriousness, is an indication to start vaccinating, even in the midst of other diseases or concurrent aggravating conditions. |
 |
Persisting in contradictions in his new conclusion, Tierney even admits to the presence of the measles disease (non-vaccine), which is highly contagious, in the Orinoco region (besides the "vaccination circle"), as well as the possibility that the virus arrived from more peripheral areas of the Yanomami territory (such as the Rio Padamo, from Chagnon's guide, mentioned above in point 6). Curiously, the mention of the measles epidemic that occurred in late 1967 in Brazil appears on the last page, but the author does not even make a link between this and the chain of contagion moving through the Yanomami territory. On the contrary, the author asserts that "cultural barriers between villages" (sic) would impede the spread of epidemics in the Yanomami area, a point that can be considered totally untrue, as demonstrated by the program of medical assistance under development in the Yanomami territory. In this regard, the members of a medical expedition conducted by the Pro-Yanomami Commission (CCPY) and by the organization Doctors Without Borders (MSF/Holland) in the region of the Siapa river basin (Venezuela) from October 1997 to May 1998 write in their report: "The eight visited communities that compose this population maintain close relations among themselves and with others located farther away in Venezuela (Platanal, Mavaca, and Mavaquita) and in Brazil (Parawau and Marari). Despite few or negligible direct contacts with our society, we are not dealing with a population that lives in a situation of epidemiological isolation in relation to other regions in the Yanomami area with older and permanent contact." 41 |
Finally, Tierney ends the text of his new conclusion of chapter 5 reinforcing his
accusations that Neel supposedly gave priority to his research objectives rather
than to the necessity of attending to the sick, and that he facilitated the spread
of the measles when his team moved through the region, accompanied by Indians
who were already infected. On these points, some further observations can be made:
| 1. |
An important new document was included in the published text (see his note 140): a research proposal presented in 1971 by Neel to Dow Chemical for testing a triple vaccine on the Yanomami. This document indirectly shows that the scientist's research proposal for the 1968 vaccination was probably to evaluate the efficacy of the vaccine, to verify the vaccine reactions, to compare the frequency of these reactions and the immunological responses to the vaccine with urban populations, and, finally, to immunize the Yanomami against measles. |
2. |
Although the various movements of the expedition might have contributed to the spread of the epidemic, we know that the traditional movements of the Yanomami among villages were already sufficient to explain its propagation from Brazil up to the Orinoco. Moreover, if measles reached the region before the team arrived, the planning and organization of their movements—whether they gave priority to either medical care or research—probably had a greater impact on the failure of the vaccination (since immunization took place later than 3 days after infection) and the lack of control over mortality (due to the ill-preparedness of the team for dealing with the serious complications of measles, mainly pneumonia), rather than on the spread of the epidemic. |
Footnotes
1* Medical report translated from Portuguese by Catherine V. Howard, Dept. of Sociology and Anthropology, Gettysburg College.
2** Participated in the Emergency Plan for Yanomami Health Care (PEASY), 1990, and in the establishment of the Yanomami Health District, Roraima (DSY/RR), 1991-1994 (clobo@hucff.ufri.br).
3 *** Developed health services in Auaris, DSY/RR, 1991.
4 Visiting researcher from the Research Institute for Development (IRD-Paris) at the Socio-Environmental Institute (ISA, Săo Paulo), specialist on the Yanomami of Brazil (BruceAlbert@aol.com).
5 Centers for Disease Control (CDC), Pink Book, chapter 9, "Measles":119-139. Available on the Internet. [requires Acrobat or other .PDF file reader]
6 ACIP, 1989, "Measles Prevention: Recommendations of the Immunization Practices Advisory Committee." MMWR 38 (S-9):1-18.
7 Redd, S. C.; L.E. Markowitz; and S. L. Katz, 1999, "Measles Vaccine." In Vaccines, ed. Plotkin and Oresten (W. B. Saunders Co.).
8 These are individuals incapable of presenting humoral immunity—by means of antibodies—or cellular immunity, due to a genetically determined deficit or, more frequently, as a secondary effect of diseases that compromise immunity or from the use of immunosuppressant agents.
9 Black, F., 1975, "Infectious Diseases in Primitive Societies." Science 187:515-518.
10 Neel, J.V.; W. R. Centerwall; and N. A. Chagnon, 1970, "Notes on the Effects of Measles and Measles Vaccine in a Virgin-Soil Population." American Journal of Epidemiology 91 (4):418-429.
11 Nutels, N., 1968, "Medical Problems of Newly Contacted Indian Groups." PAHO Scientific Publication, no.165.
12 The vaccine containing dead virus ceased being licensed in the U.S. in 1967 due to the risk of serious hypersensitive reactions upon exposure to the wild virus (Atypical Measles Syndrome), in addition to having a low efficacy and not conferring permanent immunity.
13 Hayden, G. F., 1979, "Measles Vaccine Failure." Clin. Ped.18 (3):155-167.
14 Brody, J.A. et al., 1964, "Measles Vaccine Field Trials in Alaska." JAMA 189 (5):339-342.
15 This measure also avoids superimposing the adverse effects of the vaccine with those from the disease fever, and also avoids doubts about whether a possible fever reaction originated from the vaccine or from the underlying disease.
16 Halsey, H. A.; R. Boulos; F. Mode, et al., 1985, "Response to Measles in Haitian Infants 6 to 12 Months Old: Influence of Maternal Antibodies, Malnutrition and Concurrent Illnesses." New Eng. J. Med. 313:544-549.
17 Ljungman, P., 1999, "Immunization in the Immunocompromised Host." In Vaccines, ed. Plotkin and Oresten (W. B. Saunders Co.).
18 The Declaration of Helsinki I states: "In any research on human beings, each potential subject must be adequately informed…The physician should then obtain the subject's freely-given informed consent…In the case of legal incompetence, informed consent should be obtained from the legal guardian."
(Translator's note: texts of the English versions of the Nuremberg Code and the
Declaration of Helsinki are quoted from the original documents posted at http://www.irb-irc.com/resources/nuremberg.html and http://www.irb-irc.com/resources/helsinki.html).
19 This report is posted in the site http://www.anth.uconn.edu/gradstudents/dhume/Dark/darkness/0161.htm, "The Debate over Amazon Darkness," October 17, 2000.
20 Neel, J., 1977, Ciba Foundation Symposium 49 (new series):155-177.
21 Examples can be found in: Roche et al., 1959, "Elevated Thyroidal I131 Uptake in the Absence of Goiter in Isolated Venezuelan Indians," J. Clin. Endocrin. and Metab. 19:1440; Roche et al., 1961, "Urinary Excretion of Stable Iodine in a Small Group of Isolated Venezuelan Indians," J. Clin. Endocrin. and Metab. 21:1009.
22 There are various examples of ethically dubious experiments that were supported by atomic energy commissions in the decades of the '50s and '60s, such as the radioactive exposure of American troops and mentally deficient patients in Massachusetts, conducted by the U.S. Atomic Energy Commission (see website cited above in note 16).
23 Information available on the website http://www.tamu.edu/anthropology/Lindee.html
24 Paul, Jean-Joseph et al., 1969, "A Comparison of Edmonston-B and Schwartz Measles Vaccine in Malian Children." The Lancet (March 29):665-667.
25 For this argument, Tierney relies on interview sources, which, after thirty years, are subject to memory bias; phrases lifted from articles that do not necessarily convey the general vision of the authors; and statements that preceded the official approval of the vaccine in 1963.
26 Although the author states that no other researcher used Edmonston B among indigenous peoples, there are reports even of dead virus vaccines being administered to Indians in Ecuador before an epidemic that also occurred in 1968. See Kaplan et al., 1980, "Infectious Disease Patterns in the Waorani, an Isolated Amerindian Population." Am J. Trop. Med. Hyg. 29 (2):298-312.
27 Indeed, Tierney's position conveys the prejudicial notion that the Indians are biologically fragile.
28 Among leukemia patients, pneumonia can occur with giant cells and manifestations of hemorrhaging in the absence of a rash.
29 Among the Yanomami, the average height is about 150 cm for men and 140 cm for women, and the average weight is around 45 Kg for men and 40 Kg for women. See Holmes, R., 1984, "Non-dietary Modifiers of Nutritional Status in Tropical Forest Populations of Venezuela." Interciencia 9 (5):386-391.
30 See the article by Paul, Jean-Joseph et al., 1969, cited above in note 21.
31 However, such a hypothesis may be biologically plausible, and other studies have arrived at this same conclusion. See Black, F. et al., 1969, "Measles Vaccine Reactions in a Virgin Population." Am. J. of Epidemiology 89 (2):168-175.
32 See Neel et al., 1970:421, 423.
33 O Globo (October 15, 2000), "Geneticist Researched Indians Without Authorization from FUNAI."
34 Available on the Internet at http://www.tamu.edu/anthropology/Cox.html
35 Tierney alludes to the possibility of transmission by means of trade goods, as well as to the possible existence of animal reserves for measles, which is questionable.
36 Information provided by Bruce Albert, based on interviews with the Yanomami of Toototobi and on information published by New Tribes Mission in Brown Gold (January 1968), 25 (9):3.
37 Information provided by Bruce Albert, based on a report from the Indian Protection Service (SPI), dated August 22, 1967, authored by G. Pinto Figueiredo Costa.
38 Information gathered by Bruce Albert.
39 There is no such thing as an "epidemic of bronchial pneumonia," as suggested by Tierney (bronchial pneumonia has a bacterial etiology).
40 In Tierney's own words, referring to Neel's article, "According to them (Neel and Chagnon), the Yanomami first vaccinated at Ocamo had definitive rash in strong reactions that began six days after vaccination and continued for more than 10 days (January 29-February 8)."
41 CCPY and MSF/Holland, May 1998: "Emergency Expedition to the Region of the Venezuelan Yanomami Area: Final Report."
Menu of Resources | Doctoral Program in Anthropology and History
|

 Neel indicates he was already informed of the measles epidemic
Neel indicates he was already informed of the measles epidemic