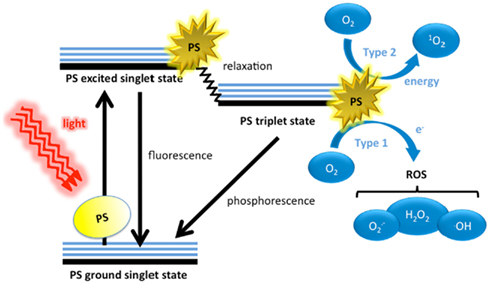
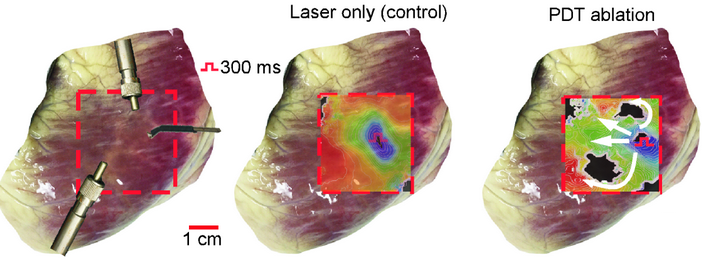
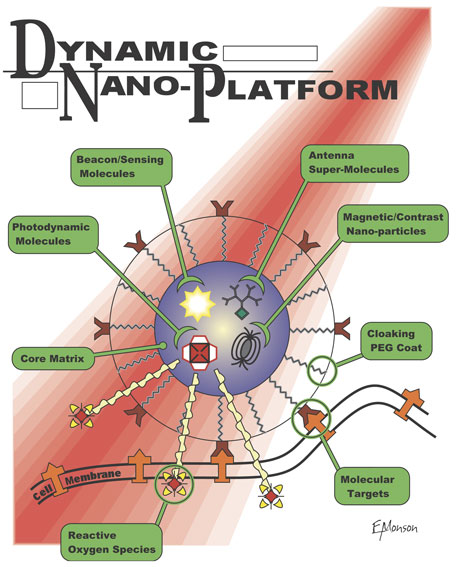
Photochemical
Nanomedicine

Chemical
Bioimaging
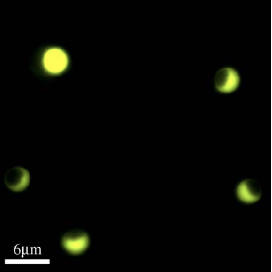
Spatially Resolved Real Time Analytical Chemistry In Vivo
Traditional analytical chemistry is performed on samples that have to be taken for analysis. For instance, blood samples taken from a patient will later undergo analysis by a lab. Very few measurements are done in real time. Exceptions are the finger monitoring of blood oxygen, or the measurement by inserted ion selective electrodes, e.g. into the brain (mainly on test animals). None of the latter measurements provide a spatial distribution of the analytes. In contrast, structural measurements provide the spatial distribution of features, and are called imaging (e.g., by using CT, MRI, or Ultrasound). Real time chemical imaging would provide much needed information to physicians. Today this is possible only with the help of a couple of highly expensive and sparsely available facilities: Functional MRI and PET (Positron Emission Tomography). Furthermore, these chemical imaging methods may require invasive probes (MRI) or the use of ionizing radiation (PET). A new approach, originated in our lab, employs photonic imaging. Notably, simple optical methods that are used in microscopy, mainly fluorescence microscopy, can only be applied "skin deep" in vivo, due to the severe light scattering by tissues.
We overcome the photon non-penetration problem by employing red or infrared laser illumination, combined with the detection of photon absorbance by the produced ultrasound; the latter easily penetrates tissues. We have demonstrated contrast nano-agents for 3-dimensional spatially resolved chemical imaging, deeply in vivo, of (1) pH (a-c) and (2) oxygen (c, d). Such pH and oxygen imaging information is of prime importance for the monitoring of cancer therapy, where the chemical nature (pH, O2) of the tumor microenvironment is highly relevant. We are working on generalizing this method for getting similar imaging information on biologically important ions (e.g., Ca, Mg, K, Na) or metabolites (e.g., glucose). Eventually, this approach could be generalized to larger molecules of interest, e.g., proteins. Notably, not even biopsies (solid or liquid) can provide such detailed information, e.g., on the spatial distribution of O2 or pH.
Selected Reading: 1. Analyst 2. Analyst 3. Nanomedicine (Lond) 4. J Biomed Opt
Cell Morphodynamics – A Cancer Lethality Fingerprinting Paradigm
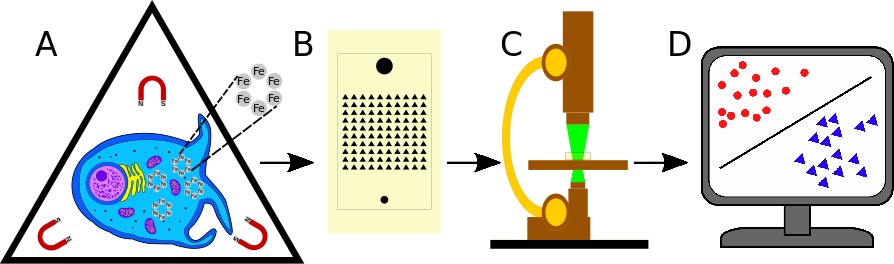
Schematic of cell morphodynamic classification. A) A captured cell expresses its morphodynamic phenotype while being gently rotated in a magnetic field, enabled by endocytosis of magnetic nanoparticles. B) The microfluidic device contains an array of triangular microwells designed so as to capture individual cells, in spaces large enough for cells to rotate freely. C) Rotating cells are fluorescently imaged on an environmentally controlled microscope stage. D) Cell images are converted by CellProfiler into parameters, used by Machine Learning algorithms to provide cellular clustering, classification, and analysis.
A cancer’s lethality is determined by its ability to spread and metastasize. Oncologists are usually unable to predict this metastatic potential by imaging a tumor or analyzing a biopsy, even if its genotype or “cell type” has been established. Is there an alternative way of enabling a timely evaluation of the cancer’s lethality, so as to guide the choice of a more, or perhaps less, drastic mode of treatment? Here we describe mathematically a new biophysical paradigm: morphodynamic cell phenotype. It characterizes a biological cell’s “shape shifting” ability and it is established by a Cell Magneto-Rotation based microfluidic imaging method. It also enables an instantaneous quantitative classification of a tumor’s metastatic potential by an Artificial Intelligence based Machine Learning algorithm. This methodology enables label-free testing of ensembles of non-fixed, minimally perturbed, free to deform, live individual cells, kept in bio- mimetic micro-environments. Our micro-fluidics based massively parallel single cell analysis assay looks into the similarities and dissimilarities of the cells’ morphological behaviors over time and we were able to identify cancer cells whose phenotype may be associated with a more, or most, malignant potential, including motility and invasiveness, thus presenting a new “Morphodynamic Histology” avenue towards the goal of precision cancer medicine.
Selected Reading: 1. PlosOne
Ratiometric Calcium Sensing Nano-PEBBLEs
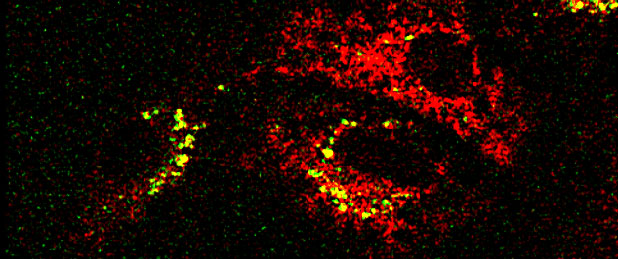
Confocal microscope image of C6 glioma (brain cancer) cells containing calcium green/sulfarhodamine nano-PEBBLEs. See Effect of Ortho-dinitro-benzene toxin moving left to right. This clarified neurotoxicological cause of brain disease found in TNT plant workers. Toxin is a side product of TNT Synthesis.
Selected Reading: 1. arXiv
Modulated Optical Nanoprobes (MOONs)

MOONs are microscopic photonic probes that look like moons: their northern hemisphere appears bright and is colored with a fluorescent indicator dye whose color reports on the local chemical environment; the other side is darkened with an opaque metal layer. They rotate in response to thermal fluctuations in a fluid (Brownian MOONs) and to rotating magnetic fields (MagMOONs), blinking as they turn through the phases of the moon. The blinking stands out against the background like a lighthouse against city lights allowing sensitive chemical analysis from microscopic probes. The probes have applications for ultrasensitive ion and small molecule sensors within cells, and robust sensitive immunoassays in blood and other fluid samples.