|


|
Hydrogenases and Alternative Energies
In order to decrease our dependency on fossil fuels, it is necessary to move to an energy economy that is based on alternatives to petroleum.
In this respect, hydrogen is the ultimate clean fuel and its use as a primary energy source desirable.[1] It is well known that hydrogen
could be produced electrochemically using energy generated from solar or biofuel cells.[2] However, current electrochemical catalysts for
H2 production suffer from inefficiency (large overpotentials) and the fact that they are based on expensive (unsustainable) platinum
catalysts.[3] In contrast, nature has solved this problem using a class of enzymes called hydrogenases.[4] The Fe-only hydrogenases are extremely
efficient in hydrogen production using a catalyst based on the cheap metal iron.[5]
In nature, three different types of transition metal-based hydrogenases are known, which catalyze the reaction:

in both directions, i.e. for hydrogen production and consumption. Of these three classes, the Fe-only hydrogenases [FeFe] are highly
evolved for catalysis and are able to produce 9000 molecules of H2 per second and per enzyme molecule at 30 oC
(Desulfovibrio desulfuricans).[6] More descriptively, one mole of [FeFe] hydrogenase could produce enough H2 in two hours
to fill the main liquid hydrogen tank of the space shuttle. Because of this outstanding catalytic rate, the Fe-only hydrogenases are of
special interest for industrial applications.
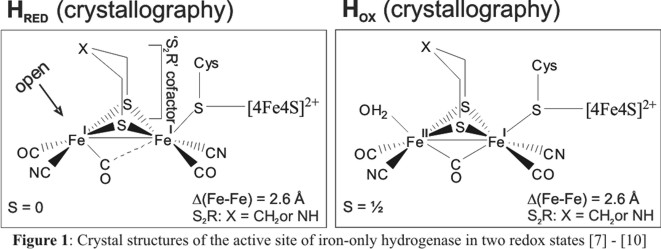
Figure 1 shows the active site of these enzymes where hydrogen formation takes place.
The catalytically active center corresponds to the Fe2 unit of the H cluster (called [FeFe]H). The underlying
[Fe2(µ-S2R)(CO)6-xLx] (L = CN-, PR3, RCN, etc.) core of the active site is
very stable and contains only the 'cheap' metal iron, which is the second reason why these systems are of such great interest for the
development of electrochemical H2-forming catalysts. Based on crystal structures of the catalytically active,
reduced Fe(I)-Fe(I) form HRED and the oxidized Fe(I)-Fe(II) form HOX of different hydrogenases [7]-[10] as well
as published spectroscopic data and density functional (DFT) calculations, the following mechanism seems plausible:
protonation of HRED leads to the formation of a hydride complex of (strictly speaking) unknown nature, which
(after one-electron transfer) is subsequently protonated leading to the generation of H2 and HOX.
Literature:
[1] Toward a Hydrogen Economy (special issue). Science 2004, 305, August 13.
[2] Vincent, K. A.; Cracknell, J. A.; Parkin, A.; Armstrong, F. A. J. Chem. Soc. Dalton Trans. 2005, 3397-3403.
[3] Turner, J. A. Science 2004, 305, 972-974.
[4] Cammack, R.; Frey, M.; Robson, R., Hydrogen as a Fuel: Learning from Nature., Taylor & Francis: London and New York, 2001.
[5] Cammack, R. Nature 1999, 397, 214-215.
[6] Hatchikian, E. C.; Forget, N.; Fernandez, V. M.; Williams, R.; Cammack, R. Eur. J. Biochem. 1992, 209, 357-365.
[7] Peters, J. W.; Lanzilotta, W. N.; Lemon, B. J.; Seefeldt, L. Science 1998, 282, 1853-1858.
[8] Nicolet, Y.; Piras, C.; Legrand, P.; Hatchikian, C. E.; Fontecilla-Camps, J. C. Structure 1999, 7, 13-23.
[9] Lemon, B. J.; Peters, J. W. Biochemistry 1999, 38, 12969-12973.
[10] Nicolet, Y.; De Lacey, A. L.; Vernede, X.; Fernandez, V. M.; Hatchikian, E. C.; Fontecilla-Camps, J. C.
J. Am. Chem. Soc. 2001, 123, 1596-1601.
|




