
The evil monkey and angry chicken were snooping around, checking out what Stewie and Overman had presented. They saw that Stewie and Overman had referenced an interesting article concerning ring-closing methathesis by a guy named Wipf:
Wipf, P.; Rector, S. R.; Takahasi, H. J. Am. Chem. Soc. 2002, 124, 14848-14849.
Overman's transformation of molecule 118 into 119 is a ring-closing metathesis that uses the second iteration of Grubbs’ catalyst as the principle reagent. The article cited here references an earlier experiment in which one of the highlights is a ring-closing metathesis reaction using a similar ruthenium-based reagent.
After seeing this, the dasterly duo of evil monkey and chicken decided to investigate this reference in the Science Citation Index to get a better idea on what Stewie and Overman were up to.
First, they noticed this article by Mardsen:
Mardsen, S. P.; McElhinney A.D. Beilstein J. Org. Chem. 2008, 4, 5397-5408.
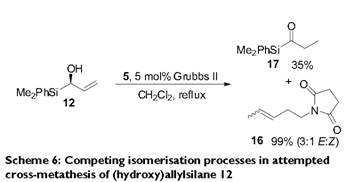
Here, the second iteration of Grubbs’ catalyst, as in the transformation between molecules 118 and 119, is employed to allow for ring closing-metathesis.
Next, the two came across this article by Arisawa:
Arisawa, M.; Terada, Y.; Takahashi, K.; Nakagawa, M.; Nishida, A. Chem. Rec. 2007, 7, 238-253.
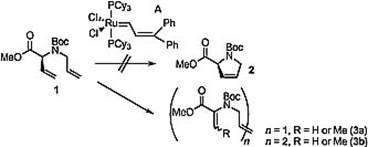
Here, the first generation of Grubbs’ catalyst is used to allow for a ring-closing metathesis.
Last, the monkey and chicken found this article by Barberis:
Barberis, M.; Garcia-Losada, P.; Pleite, S.; Rodriguez J. R.; Sorian, J. F.; Mendiola J. Tetrahedron Lett. 2005, 46, 4847-4850.
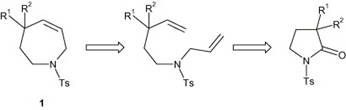
Here, an azepine ring undergoes ring-closing metathesis as seen in the Wipf article cited by Overman (and Stewie).
