19 January 2014
Bd in Bahia and Espirito Santo
Jan/22/14 11:24
Jan. 22, 2014.
The Bd in Brazil crew is back again this year, this time moving the transect north into the states Espirito Santo and Bahia of the Atlantic Forest. We are a team of mycologists and herpetologists interested in understanding the biology of a fungus disease that globally is decimating amphibian populations (Wake and Vredenburg 2008). The ultimate goal of the Brazilian research is to sample across the remaining fragments of the Brazilian Atlantic Forest from Rio Grande do Norte to Rio Grande do Sul for molecular epidemiological studies of the fungus.
This year’s crew is again a combination of researchers at UNICAMP (Felipe Toledo Lab, Campinas, Brasil) and University of Michigan (Tim James Lab) and Cornell University (Kelly Zamudio Lab). We also had the pleasure of adding a member from UFAL (Universidade Federal de Alagoas), Anyelet Valencia from Tamí Mott’s lab.
In brief, we found Bd at 4/5 sites in tadpoles, with prevalence ranging from very high (50%) to very low (1%) on the basis of morphological examination of tadpoles. The three sites with high levels of Bd were all high elevation (>600 m) localities, whereas the lowland rainforest showed a near absence of Bd in tadpoles. It will be of great interest to compare the results we have on Bd prevalence from the larval data to that of the adults. At each of the 5 localities, over 100 adults were swabbed and analysis of Bd infection by qPCR is in process at UNICAMP. At one locality near Pedra Azul, we collected a second year North American bullfrog tadpole, infected with Bd, in among a diverse pool of native tadpoles, some also infected.

A couple tadpoles from the pond near Pedra Azul, Espirito Santo.
What is becoming abundantly clear is that there is a strong correlation with detectable Bd infection of larvae and temperature of the water from which they are collected. Cool streams running down from high elevation sites seem to invariably yield infected tadpoles, whereas warm unshaded ponds as found in deforested sites with daily maximum temperature above 25 have all uninfected larvae. Of course this fits in very well with what we know about the tolerance of Bd in the laboratory. That is, growth is essentially halted when Bd experiences temperatures at or above 28 C (Piotrowski et al. 2004). On the other hand, adults collected from the same warm ponds can be quite infected with Bd. These observations of differential infection of adults and larvae prompt at least a couple questions regarding the disease dynamics. Firstly, how do the adults become infected in the first place? Where is Bd lurking among the places to which adults might journey? Are adults acquiring their infections by crossing these Bd-infested streams or is there another refuge for the fungus in these warmer sites? Secondly, what effect does this high level of Bd infection in tadpoles have on the adults of riparian species of frogs? Are there sub-lethal effects or mortalities that have gone undetected.

Dendropsophus branneri from southern Bahia. These frogs breed in permanent and vernal pools and live in a variety of habitats except closed canopy forest. Bd infection status is unknown at present.
The answer to this second question is critical to our understanding of the impacts of Bd on Brazilian amphibians. While the consensus, or absence of evidence, points to no known Bd-related declines in the Atlantic Forest of Brazil, it is a statement which largely speaks to poor data. Also, Bd could also have large impacts on population density without obvious extinctions and observed die offs. As it stands, it is an interesting correlation that the 31 species suspected of undergoing declines are enriched in those who inhabit high elevation riparian sites (Eterovick et al. 2005). Our crew has now visited a couple sites where declines are reported (PARNASO, Teresopolis, RJ and Santa Teresa, ES) and these two localities are sites where Bd occurs at high prevalence.
Our sampling has now expanded over a large portion of the Atlantic Forest and has sadly extended the known distribution of Bd into additional states, Bahia and Espirto Santo. The once continuous Atlantic Coastal Forest of Brazil appears to have Bd wherever conditions are favorable for the fungus. Interestingly, there appears to be a negative correlation between Bd prevalence and degree of forest fragmentation (Becker and Zamudio, 2011). This paradox has been explained in a couple ways, either the degraded habitat selects for weedy species of frogs that thrive in disturbed areas, and there may be a correlation between aggressive weedy species and resistance to Bd, or the reduction in diversity may reduce the equilibrium infection frequency of Bd in a community. Alternatively the increased fragmentation leads to increased solar radiation and thus higher temperatures, making water and terrestrial habitats perhaps less favorable to Bd.
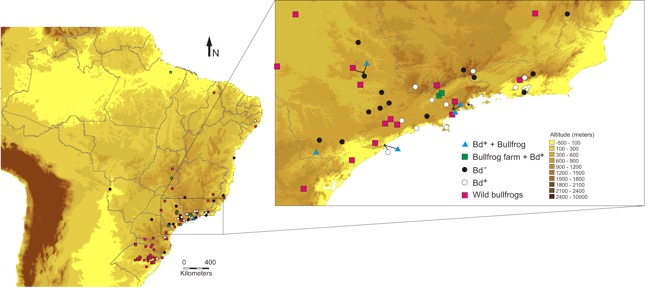
Map of the distribution of Bd in Brazil as known in 2012 (Fig 7 of Schloegel et al. 2012).
Despite the observation that Bd may occur in lower frequencies in deforested sites, stopping or reversing deforestation in Brazil should be a central priority of conservation biologists working here. Though not based on any specific data or experiments, my impression is that the the Atlantic Forest and its biodiversity could rebound if the deforestated areas are allowed to recover, a change in land use policies is implemented, and enough time elapses. While settlement contributes a large proportion to the loss of forest, obvious are also the hectares infinitos devoted to raising cattle, growing eucalyptus, sugarcane, coffee, soy, bananas, and etc. With a Brazilian economy emerging rapidly, including a burgeoning middle class with US sized consumerism, it will require progressive leadership to value future resources over money in pockets. Nevertheless, the people are concentrated into urban environments, with a rural landscape that is deforested primarily for agriculture or logging. When I look at this fragmented landscape, I see potential for regrowth of the forest, for the salvation of the wildlife and plant species and their diversity. What is left must be protected at all costs because extinctions are much more likely given small fragment sizes and invasive species. What has been razed for cattle and sugarcane must be allowed to grow back by a few centuries of abandon. I remain hopeful.
Back to our research. Brazil is known to harbor three genotypes of Bd: Bd-GPL (a global genotype associated with all known Bd related declines), Bd-Brazil (a genotype known only from Brazil and possibly Japan with unknown virulence), and Bd-Hybrid (sexual genotypes produced between Bd-GPL and Bd-Brazil). We are attempting to use our cultures of Bd to understand how temporal and spatial patterns of genotypes can answer the question of what and when Bd came to Brazil. We predicted last year that Teresopolis, where declines are recorded, would have the hyper virulent Bd-GPL genotype, and that held true (in prep). We predict the same for Santa Teresa, ES, where species like Colostethus olfersioides are in decline (Weygoldt 1989; Stud. Neotrop. Fauna Environ. 243: 249). As part of the same hypothesis we predicted that Bd-Brazil was endemic, perhaps extending deep into the time when there were multiple, isolated refugia for fauna during Pleistocene global cooling and drying events. Maybe there are more genotypes of Bd in the Atlantic Forest that have yet to be recovered centered around these refugia. In frogs at least, there are known to be three refugia in the AF relating to concentrated forest fragments (Carnaval et al. 2009). Maybe the presence of these endemic genotypes has provided some sort of defense against Bd-GPL, some sort of immunization or competition among the global hyper virulent lineage? Currently, we have found Bd-GPL at every site we have sampled, but Bd-Brazil only at sites in Paraná, Santa Catarina, and São Paulo. This would seem to be consistent with a model of Bd-GPL recently replacing Bd-Brazil, perhaps through their introduction on bullfrogs, which arrived in Brazil in the 1930’s. However, this hypothesis is challenged by the recent observation that both Bd-Brazil and Bd-GPL genotypes have been detected from museum specimens dating back to at least 1916 (Rodriguez et al. 2014). Bd has apparently been in the Atlantic Forest since 1894.


Proceratophrys schirchi. These Leptodactylids are able to defend themselves from predation by inflation. Photos: T. James (Top), K. R. Zamudio (bottom).
Getting to the bottom of the genotype mystery will require integrated studies of museum collections, population genetics, and infection experiments. Detecting population expansion could use coalescent methods in population genetics, but a general lack of variation and polyploidy has been a big hindrance. We are currently developing new markers to ask the question of whether the Bd-Brazil or the Bd-GPL strains have greater heterozygosity in the AF. That seems to have been the case based on genome resequencing (Rosenblum et al. 2013), but many more isolates need to be tested. I dream of establishing long term monitoring plots in the area we know that all three genotypes occur so that the dynamics of the genotypes in time and space can be studied properly.
Até mais!
The Bd in Brazil crew is back again this year, this time moving the transect north into the states Espirito Santo and Bahia of the Atlantic Forest. We are a team of mycologists and herpetologists interested in understanding the biology of a fungus disease that globally is decimating amphibian populations (Wake and Vredenburg 2008). The ultimate goal of the Brazilian research is to sample across the remaining fragments of the Brazilian Atlantic Forest from Rio Grande do Norte to Rio Grande do Sul for molecular epidemiological studies of the fungus.
This year’s crew is again a combination of researchers at UNICAMP (Felipe Toledo Lab, Campinas, Brasil) and University of Michigan (Tim James Lab) and Cornell University (Kelly Zamudio Lab). We also had the pleasure of adding a member from UFAL (Universidade Federal de Alagoas), Anyelet Valencia from Tamí Mott’s lab.
In brief, we found Bd at 4/5 sites in tadpoles, with prevalence ranging from very high (50%) to very low (1%) on the basis of morphological examination of tadpoles. The three sites with high levels of Bd were all high elevation (>600 m) localities, whereas the lowland rainforest showed a near absence of Bd in tadpoles. It will be of great interest to compare the results we have on Bd prevalence from the larval data to that of the adults. At each of the 5 localities, over 100 adults were swabbed and analysis of Bd infection by qPCR is in process at UNICAMP. At one locality near Pedra Azul, we collected a second year North American bullfrog tadpole, infected with Bd, in among a diverse pool of native tadpoles, some also infected.

A couple tadpoles from the pond near Pedra Azul, Espirito Santo.
What is becoming abundantly clear is that there is a strong correlation with detectable Bd infection of larvae and temperature of the water from which they are collected. Cool streams running down from high elevation sites seem to invariably yield infected tadpoles, whereas warm unshaded ponds as found in deforested sites with daily maximum temperature above 25 have all uninfected larvae. Of course this fits in very well with what we know about the tolerance of Bd in the laboratory. That is, growth is essentially halted when Bd experiences temperatures at or above 28 C (Piotrowski et al. 2004). On the other hand, adults collected from the same warm ponds can be quite infected with Bd. These observations of differential infection of adults and larvae prompt at least a couple questions regarding the disease dynamics. Firstly, how do the adults become infected in the first place? Where is Bd lurking among the places to which adults might journey? Are adults acquiring their infections by crossing these Bd-infested streams or is there another refuge for the fungus in these warmer sites? Secondly, what effect does this high level of Bd infection in tadpoles have on the adults of riparian species of frogs? Are there sub-lethal effects or mortalities that have gone undetected.

Dendropsophus branneri from southern Bahia. These frogs breed in permanent and vernal pools and live in a variety of habitats except closed canopy forest. Bd infection status is unknown at present.
The answer to this second question is critical to our understanding of the impacts of Bd on Brazilian amphibians. While the consensus, or absence of evidence, points to no known Bd-related declines in the Atlantic Forest of Brazil, it is a statement which largely speaks to poor data. Also, Bd could also have large impacts on population density without obvious extinctions and observed die offs. As it stands, it is an interesting correlation that the 31 species suspected of undergoing declines are enriched in those who inhabit high elevation riparian sites (Eterovick et al. 2005). Our crew has now visited a couple sites where declines are reported (PARNASO, Teresopolis, RJ and Santa Teresa, ES) and these two localities are sites where Bd occurs at high prevalence.
Our sampling has now expanded over a large portion of the Atlantic Forest and has sadly extended the known distribution of Bd into additional states, Bahia and Espirto Santo. The once continuous Atlantic Coastal Forest of Brazil appears to have Bd wherever conditions are favorable for the fungus. Interestingly, there appears to be a negative correlation between Bd prevalence and degree of forest fragmentation (Becker and Zamudio, 2011). This paradox has been explained in a couple ways, either the degraded habitat selects for weedy species of frogs that thrive in disturbed areas, and there may be a correlation between aggressive weedy species and resistance to Bd, or the reduction in diversity may reduce the equilibrium infection frequency of Bd in a community. Alternatively the increased fragmentation leads to increased solar radiation and thus higher temperatures, making water and terrestrial habitats perhaps less favorable to Bd.

Map of the distribution of Bd in Brazil as known in 2012 (Fig 7 of Schloegel et al. 2012).
Despite the observation that Bd may occur in lower frequencies in deforested sites, stopping or reversing deforestation in Brazil should be a central priority of conservation biologists working here. Though not based on any specific data or experiments, my impression is that the the Atlantic Forest and its biodiversity could rebound if the deforestated areas are allowed to recover, a change in land use policies is implemented, and enough time elapses. While settlement contributes a large proportion to the loss of forest, obvious are also the hectares infinitos devoted to raising cattle, growing eucalyptus, sugarcane, coffee, soy, bananas, and etc. With a Brazilian economy emerging rapidly, including a burgeoning middle class with US sized consumerism, it will require progressive leadership to value future resources over money in pockets. Nevertheless, the people are concentrated into urban environments, with a rural landscape that is deforested primarily for agriculture or logging. When I look at this fragmented landscape, I see potential for regrowth of the forest, for the salvation of the wildlife and plant species and their diversity. What is left must be protected at all costs because extinctions are much more likely given small fragment sizes and invasive species. What has been razed for cattle and sugarcane must be allowed to grow back by a few centuries of abandon. I remain hopeful.
Back to our research. Brazil is known to harbor three genotypes of Bd: Bd-GPL (a global genotype associated with all known Bd related declines), Bd-Brazil (a genotype known only from Brazil and possibly Japan with unknown virulence), and Bd-Hybrid (sexual genotypes produced between Bd-GPL and Bd-Brazil). We are attempting to use our cultures of Bd to understand how temporal and spatial patterns of genotypes can answer the question of what and when Bd came to Brazil. We predicted last year that Teresopolis, where declines are recorded, would have the hyper virulent Bd-GPL genotype, and that held true (in prep). We predict the same for Santa Teresa, ES, where species like Colostethus olfersioides are in decline (Weygoldt 1989; Stud. Neotrop. Fauna Environ. 243: 249). As part of the same hypothesis we predicted that Bd-Brazil was endemic, perhaps extending deep into the time when there were multiple, isolated refugia for fauna during Pleistocene global cooling and drying events. Maybe there are more genotypes of Bd in the Atlantic Forest that have yet to be recovered centered around these refugia. In frogs at least, there are known to be three refugia in the AF relating to concentrated forest fragments (Carnaval et al. 2009). Maybe the presence of these endemic genotypes has provided some sort of defense against Bd-GPL, some sort of immunization or competition among the global hyper virulent lineage? Currently, we have found Bd-GPL at every site we have sampled, but Bd-Brazil only at sites in Paraná, Santa Catarina, and São Paulo. This would seem to be consistent with a model of Bd-GPL recently replacing Bd-Brazil, perhaps through their introduction on bullfrogs, which arrived in Brazil in the 1930’s. However, this hypothesis is challenged by the recent observation that both Bd-Brazil and Bd-GPL genotypes have been detected from museum specimens dating back to at least 1916 (Rodriguez et al. 2014). Bd has apparently been in the Atlantic Forest since 1894.


Proceratophrys schirchi. These Leptodactylids are able to defend themselves from predation by inflation. Photos: T. James (Top), K. R. Zamudio (bottom).
Getting to the bottom of the genotype mystery will require integrated studies of museum collections, population genetics, and infection experiments. Detecting population expansion could use coalescent methods in population genetics, but a general lack of variation and polyploidy has been a big hindrance. We are currently developing new markers to ask the question of whether the Bd-Brazil or the Bd-GPL strains have greater heterozygosity in the AF. That seems to have been the case based on genome resequencing (Rosenblum et al. 2013), but many more isolates need to be tested. I dream of establishing long term monitoring plots in the area we know that all three genotypes occur so that the dynamics of the genotypes in time and space can be studied properly.
Até mais!
